|
Isotopes are very similar versions of the same element. Finally, multiply the two numbers together to get the average atomic mass of an element. Next, calculate the percent natural abundance of each isotope. 
First, find the atomic mass of all the stable isotopes of the element. There are a few steps involved in calculating the average atomic mass of an element. However, for most elements, there are several naturally occurring isotopes that can be used to determine the average atomic mass. For heavier elements, the binding energy of the nucleus can cause a slight change in mass. Some of the lighter elements in the periodic table have only two protons in the nucleus, while others have one or more. It can be determined by looking at the periodic table. 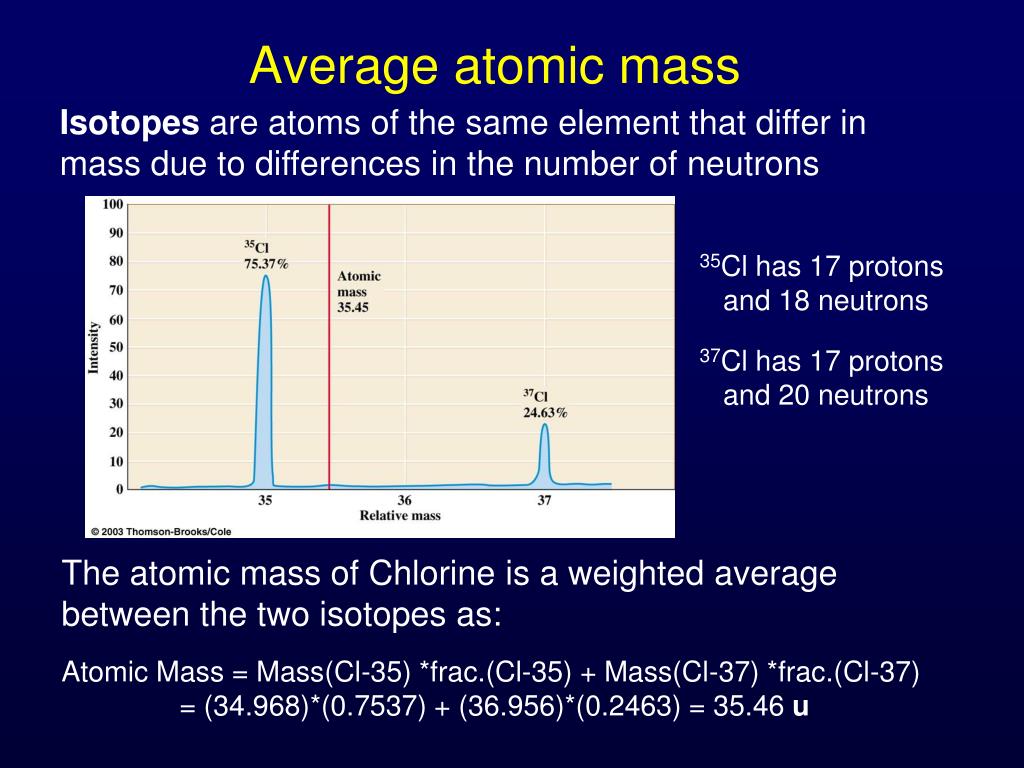
The number of neutrons in the nucleus is usually very close to the number of protons. The atomic mass of an element is a number of protons and neutrons in the nucleus. This is done by taking the natural abundance of the isotopes and multiplying it by the isotope’s atomic mass. One is to find the average of the atomic mass of all the isotopes of an element. 
If you want to calculate the average atomic mass of an element, there are a few different ways to do it. How to Calculate Average Atomic Mass of an Elements This method will allow you to get a more accurate result. The first way to do this is to use the weighted average method. If you are trying to find the average atomic mass of an element, there are several ways to calculate it.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
